Caspase 3, Human Recombinant
General description
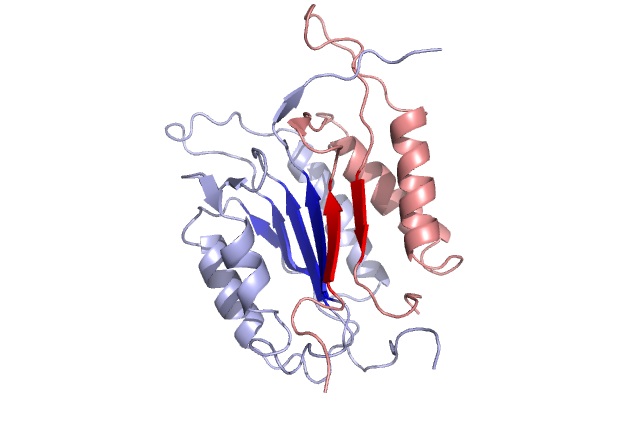
Caspase-3 (also known as CPP32, Yama and apopain) is a major member of the caspase-family of cysteine proteases. Caspase-3 exists in cells as an inactive 32 kDa proenzyme. During apoptosis Procaspase-3 is processed at aspartate residues by self-proteolysis and/or cleavage by upstream caspases, such as Caspase-6 (Mch2), Caspase-8 (Flice) and grazyme B. The processed form of Caspase-3 consists of large (17 kDa) and small (11 kDa) subunits which associate to form the active enzyme. The active Caspase-3 has been shown involving in the proteolysis of several important molecules, such as poly (ADP-ribose) polymerase (PARP), the sterol regulatory element binding proteins (SREBPs), focal adhesion kinase (FAK), and others. The recombinant active human Caspase-3 expressed in E. coli spontaneously undergoes auto-processing to yield subunits characteristic of the native enzyme (Full length gene Accession No. NP_004337). The active Caspase-3 preferentially cleaves Caspase-3 substrates (e.g., DEVD-AFC or DEVD-pNA).
Synonyms: Caspase-3, CASP-3, Apopain, Cysteine protease CPP32, CPP-32, Protein Yama, SREBP cleavage activity 1, SCA-1
Gene Symbol: CASP3
Gene ID: 836
Source: E. coli.
Form: Semi-Dry
Molecular Weight: Large (17 kD) and small (11 kD) subunits
Reconstitution Inst: Reconstitute in water
Handling: Centrifuge the vial prior to opening
Storage temp.: Shipped at -20°C. For long term storage store at -70°C