The host cell extracts for Histidine-Tagged (His-Tag) protein contain a number of endogenous proteases, that are capable of modifying the proteins present in the extract/lysate. The need for protease inhibitors arises to protect the proteins from damage caused by these proteases released during the cellular lysis. Also, to improve the yield of native proteins, use of protease inhibitor cocktail along with phosphatase and other inhibitors is recommended during the extraction process.
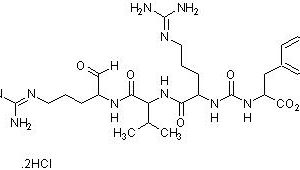
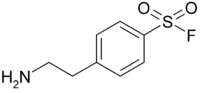
Our Protease Inhibitor Cocktail, His-Tag [100X] has a broad specificity for the inhibition of serine, cysteine, acid, thermolysin-like proteases and aminopeptidases and metalloproteases. It contains optimized concentration of protease inhibitors: AEBSF (4-[2-Aminoethyl] benzenesulfonyl fluoride hydrochloride), Bestatin, Pepstatin A, E-64 (N-[trans-Epoxysuccinyl]-L-leucine 4-guanidinobutylamide), Phosphoramidon protease inhibitors and other proprietary component(s) for broad spectrum inhibition of proteases that target serine proteases (e.g., trypsin, chymotrypsin, plasmin, kallikrein, and thrombin), cysteine proteases (e.g., calpain, papain, cathepsin B, and cathepsin L), aminopeptidases (e.g., leucine aminopeptidase and alanyl aminopeptidase, acid proteases (e.g., pepsin, rennin, and cathepsin D, and many microbial aspartic proteases), and membrane metallo-endopeptidase (e.g., thermolysin and collagenase), etc. The inhibitor cocktail does not contain any chelators, such as EDTA, to optimize its use in the purification of histidine-tagged proteins and is compatible with immobilized metal affinity chromatography.
The inhibitor cocktail is supplied as a ready to use solution in DMSO, at a 100X concentration, which makes it easier to use at lower volumes at 1X, 2X or more for samples with high protease activities. The supplied inhibitor cocktail helps in preserving the proteins from degradation by proteases and can be used with the lysis buffer for His-Tag protein extractions or lysate.








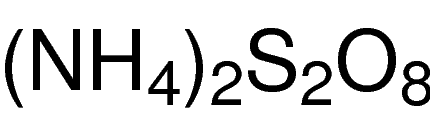
![IEF Anode Buffer [50X]](https://www.cephamls.com/wp-content/uploads/2019/02/10496-3-430x334.jpg)