Proteomics is the large-scale study of proteins which involves everything from examining their structure to trying to understand more about their physiological role or functions. First coined in 1995, the term ‘proteomics’ was used to describe the large-scale characterization of all proteins found in a cell, tissue, or organ. Since then, advancing technologies has enabled proteomics to evolve and proteomes can now be studied in great depth. Proteomics has a wide range of applications. It is crucial in early disease diagnosis and monitoring disease progression. In addition, it also plays a vital role in drug development as a target molecule.
Why study proteomes?
Proteins carry out a wide range of vital functions within our cells and tissues. Understanding what proteins are being translated can help us to determine the body’s response to specific changes or provide us with more information about the mechanisms involved in molecular pathways. The proteome of a cell or tissue is not constant, changing from cell to cell, over time, and in response to external stimuli.
Proteomics enables us to study the set of proteins that are being produced at a given time and to some degree often reflects the underlying transcriptome. However, protein activity in eukaryotic cells can be complex and their levels are not only dependent on mRNA levels but also translational control and regulation mechanisms. As a result, proteomics carried out in conjunction with transcriptomics can help us understand gene function but only proteomics can provide us with a data set that is most relevant to characterizing how a biological system function.
Proteins undergo post-translational checks and modifications and so don’t always reflect what is happening at the transcriptome level. As a result, studying the proteome can be more relevant for understanding how biological systems are functioning (Kep17 via Wikimedia commons).
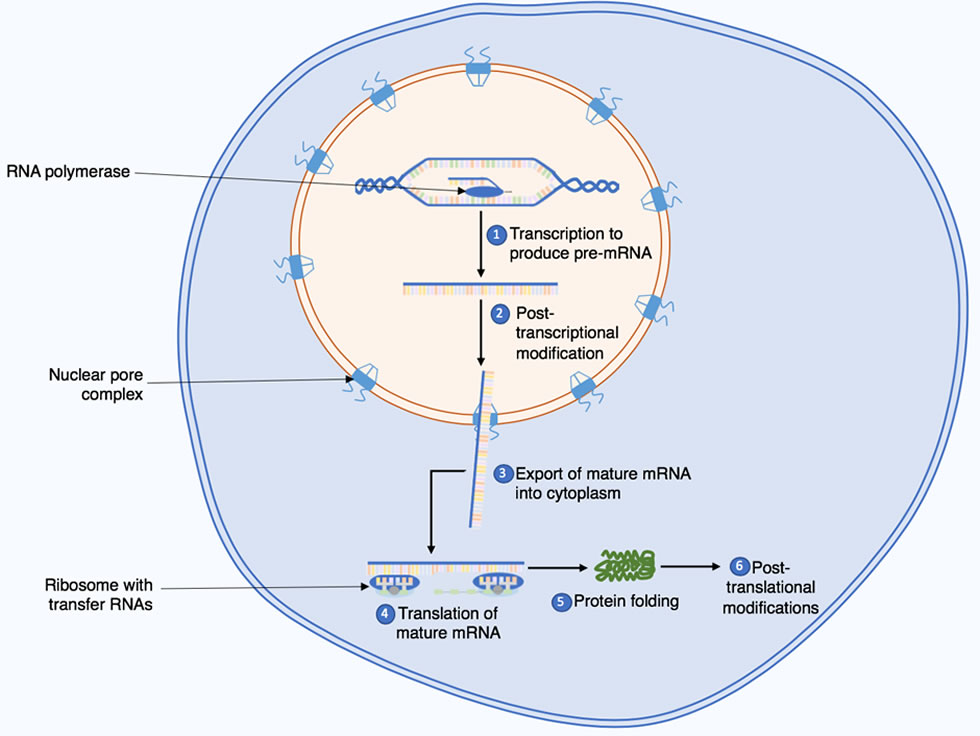
Proteomics has a wide range of applications and in addition to the applications previously mentioned, it can also be used to investigate:
- protein expression within a cell including where they are expressed and under what conditions
- rates of protein production, degradation, and their steady-state abundance
- protein modifications such as post-translational modifications
- the movement of proteins between different subcellular compartments
- the involvement of proteins in metabolic pathways
- protein-protein interactions
Techniques used in proteomic studies
Traditionally, three main methods are used to separate complex protein or peptide samples: denaturing polyacrylamide gel electrophoresis (PAGE) or sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE), two-dimensional gel electrophoresis, and high-performance liquid chromatography (HPLC).
SDS-PAGE is the most widely used protein separation technique because it is simple, cost-effective, and has a high level of reproducibility. This method works by separating proteins based on their primary structure or size and can be used to estimate molecular weights for unknown proteins and to check the purity of a sample. Two-dimensional (2-D) gel electrophoresis adds an extra step by applying isoelectric focusing (IEF) to separate proteins according to their isoelectric point before carrying out SDS-PAGE. This enables thousands of proteins to be separated. 2-D gel electrophoresis can also easily interface with immunoblotting techniques. The third technique is HPLC which is an advanced form of column chromatography and is used for the analysis of a vast array of organic molecules and ions. This separation technique uses pumping systems, separation columns, and detectors to help identify and quantify compounds.
The advancement of proteomic technologies
More modern technologies are also used in proteomics. Mass spectrometry is an analytical tool that is useful for measuring the mass-to-charge ratio of the molecules present in a sample. These measurements can then be used to calculate the exact molecular weight of these molecules. Mass spectrometers can also be used to determine the structure and chemical properties of the molecules in question. This technique has become a core technology in proteomics. Other techniques that are commonly used included electrospray ionization and matrix-assisted laser desorption/ionization.
Another technology that has enabled rapid progression in proteomics is the protein microarray or protein chip. This high-throughput technique uses multiplex assays to characterize proteins, determining their function, interactions, and activities. Protein microarrays enable large numbers of proteins to be studied in parallel and can also be used to examine peptides and other low molecular weight compounds. It works by using capture proteins bound to a support surface, such as a glass slide, bead, or nitrocellulose membrane. Once the sample has been added, a probe is used. This probe is often labelled with a fluorescent dye and emits a fluorescent signal once a reaction has occurred between the probe and an immobilised protein. This signal is read by a laser scanner. This highly sensitive technique is both rapid and economical, using only small quantities of samples and reagents.
How We Can Help
Cepham Life Science offers a wide range of products for use in proteomic studies, from buffers, columns, and protein extraction kits to digestion kits suitable for use with mass spectroscopy. For more information on our range of products have a look at our website or contact one of our team who will be happy to answer all your questions.
Contact Cepham Life Sciences Today!
Cepham Life Sciences team is available to meet all your proteomics and molecular biology laboratory needs. We have a wide range of quality research materials, and our team of experts are on hand to offer advice and answer any questions that you have. You can reach us by phone at (410) 636-4954 or Toll Free 1-800-257-1565 (USA/CANADA) or by email.
Follow us on Twitter, Facebook, Google+, and LinkedIn to keep up to date on all of our products and promotions.





