General Description
In protein biology research, affinity purification of His-tagged fusion proteins is the most common application for metal-chelates. Cobalt metals immobilized by NTA-chelation are the routine systems of choice for this application. In appropriate columns or cartridge units, such resins provide for purification of ~1 to 80 milligrams of His-tagged protein per milliliter of agarose beads.
The chelators most commonly used as ligands for IMAC are nitrilotriacetic acid (NTA) and iminodiacetic acid (IDA). NTA-agarose resin is prepared, it can be efficiently “loaded” with the desired divalent metal (e.g., Ni, Co, Cu, and Fe) and the resulting affinity support is usually called Cobalt-chelate or Co-NTA resin. The particular metal and chelation chemistry of a support is imperative to determine its binding properties and suitability for specific applications of IMAC.
Cobalt-NTA Agarose Resin is used for preparative purification of histidine-tagged recombinant proteins from all prokaryotic and eukaryotic expression systems. Cobalt has a binding capacity of >25 mg/ml with approximately 60 kDa protein and binds fewer host protein contaminants, resulting in lower background than nickel resins without metal contamination in eluted histidine-tagged protein sample. Since cobalt exhibits a more specific interaction with histidine tags resulting in less nonspecific interaction, it is the preferred divalent cation for purifying His-tagged proteins where high purity is a primary concern. It readily allows for protein purification under native or denaturing conditions. Cobalt Agarose system is compatible with Ni/Cu/Co Resin Wash Buffer and Histidine Protein Elution Buffer.
Our Cobalt-NTA Agarose Resin consists of highly cross-linked 6% agarose with an immobilized chelating group. The talon ligand is a tetra-dentate chelator charged with cobalt.
Features:
- Ready-to-use IMAC resin that utilizes Cobalt for purifying 6x His-tagged proteins
- High selectivity: appropriate for valuable recombinant proteins in limited quantities
- Allows for purification of proteins under native or denaturing conditions




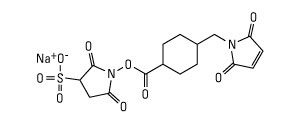

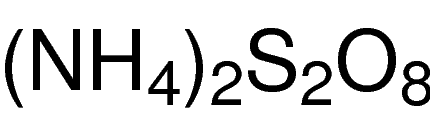



![IEF Anode Buffer [50X]](https://www.cephamls.com/wp-content/uploads/2019/02/10496-3-430x334.jpg)
![IEF Cathode Buffer (pH 3-7) [10X]](https://www.cephamls.com/wp-content/uploads/2019/02/10498-3-430x334.jpg)