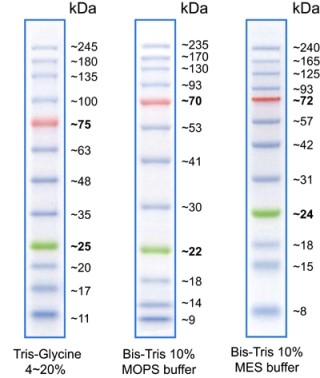
General Description
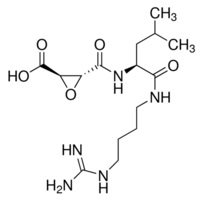
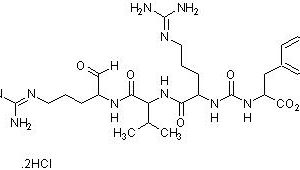
E-64 is an irreversible, cell permeable and potent inhibitor of cysteine proteases, while being highly selective. It is unreactive with the functional thiol group of non-protease enzymes, such as L-lactate dehydrogenase or creatine kinase, and does not inhibit serine proteases, except trypsin, like other cysteine protease inhibitors leupeptin and antipain. E-64 irreversibly binds to an active thiol group by its trans-epoxysuccinyl group (active moiety) to form a thioether linkage, in many cysteine proteases and inhibits calpain, papain, and cathepsin B, cathepsin L and has been reported to inhibit intracellular Bax protease activity, and reduce oxidative stress, which includes a decrease in MDA levels, ICAM-1 expression, and MOP activity.
E-64 is a very useful cysteine protease inhibitor for use in vivo since it has a specific inhibitory potential, is permeable in cells and tissues, and has low toxicity.
Solubility: DMSO
Storage: 2-8o C








![IEF Anode Buffer [50X]](https://www.cephamls.com/wp-content/uploads/2019/02/10496-3-430x334.jpg)
![IEF Cathode Buffer (pH 3-7) [10X]](https://www.cephamls.com/wp-content/uploads/2019/02/10498-3-430x334.jpg)