General Description
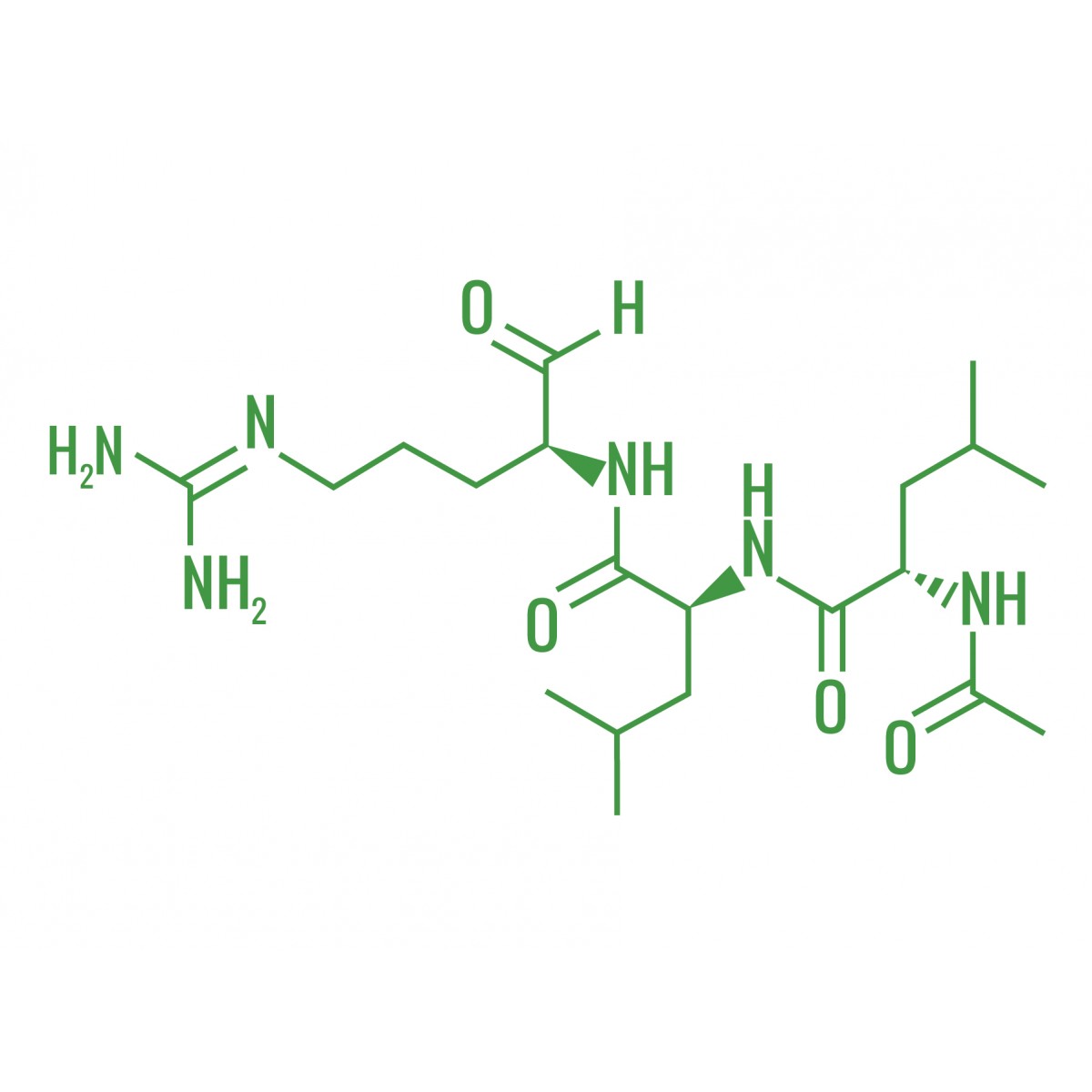
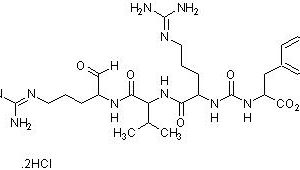
Leupeptin is a reversible competitive protease inhibitor which inhibits serine and cysteine proteases including trypsin, plasmin, calpain, papain, and cathepsins B, H and L. It does not inhibit pepsin, cathepsins A and D, thrombin, or α-chymotrypsin. Since leupeptin contains an aldehyde group in its structure, it may act as a reducing agent to cause interference in the protein estimation with Lowry method.
Leupeptin helps to protect motor neurons from apoptosis. It also helps to enhance muscle function following nerve injury. It possesses neurotrophic action and has a growth-stimulating effect.
Application:
Leupeptin has been used as a protease inhibitor:
- in ice-cold lysis buffer to harvest cells for western immunoblotting and immunoprecipitation
- in chromatin immunoprecipitation (ChIP) lysis buffer for the isolation of fragmented chromatin samples
- in lysis buffer I to lyse the cells for tandem affinity purification (TAP)
There have been numerous studies using leupeptin to protect against hearing loss caused by acoustic over-stimulation or the ototoxic antibiotic gentamicin. (Loss of cochlear hair cells is believed to be mediated by calpain)
Working Concentration: 10-100 μM.
Solubility: Soluble in Water
Storage: -20 °C




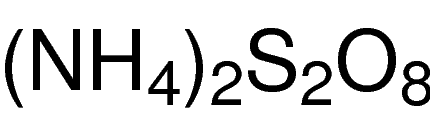

![IEF Anode Buffer [50X]](https://www.cephamls.com/wp-content/uploads/2019/02/10496-3-430x334.jpg)
![IEF Cathode Buffer (pH 3-7) [10X]](https://www.cephamls.com/wp-content/uploads/2019/02/10498-3-430x334.jpg)






