General description
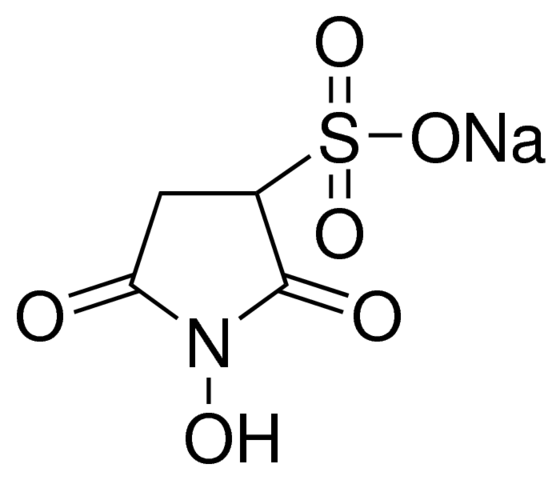
N-hydroxysulfosuccinimide sodium salt is a water-soluble sulfonated analog of N-hydroxysuccinimide. It can undergo a coupling reaction with carboxylic acids in the presence of carbodiimides such as 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC) to form hydrophilic N-hydroxysulfosuccinimide active esters that are useful in protein cross-linking studies. It enables control and modification of carbodiimide crosslinking reactions involving activation of carboxylates (—COOH) for conjugation with primary amines (—NH2). Derivatives are easily synthesized by mixing the Sulfo-NHS with a carboxyl-containing molecule and a dehydrating agent such as the carbodiimide EDC (EDAC). The method is the basis for generating multiple protein labeling reagents, including amine-reactive fluorescent dyes, biotin affinity tags and pegylation compounds.
Applications:
- improve efficiency of EDC coupling reactions
- convert carboxyls to amine-reactive Sulfo-NHS esters
- crosslink proteins to carboxyl-coated beads or surfaces more efficiently
- activate nanoparticles with amine-reactive Sulfo-NHS esters